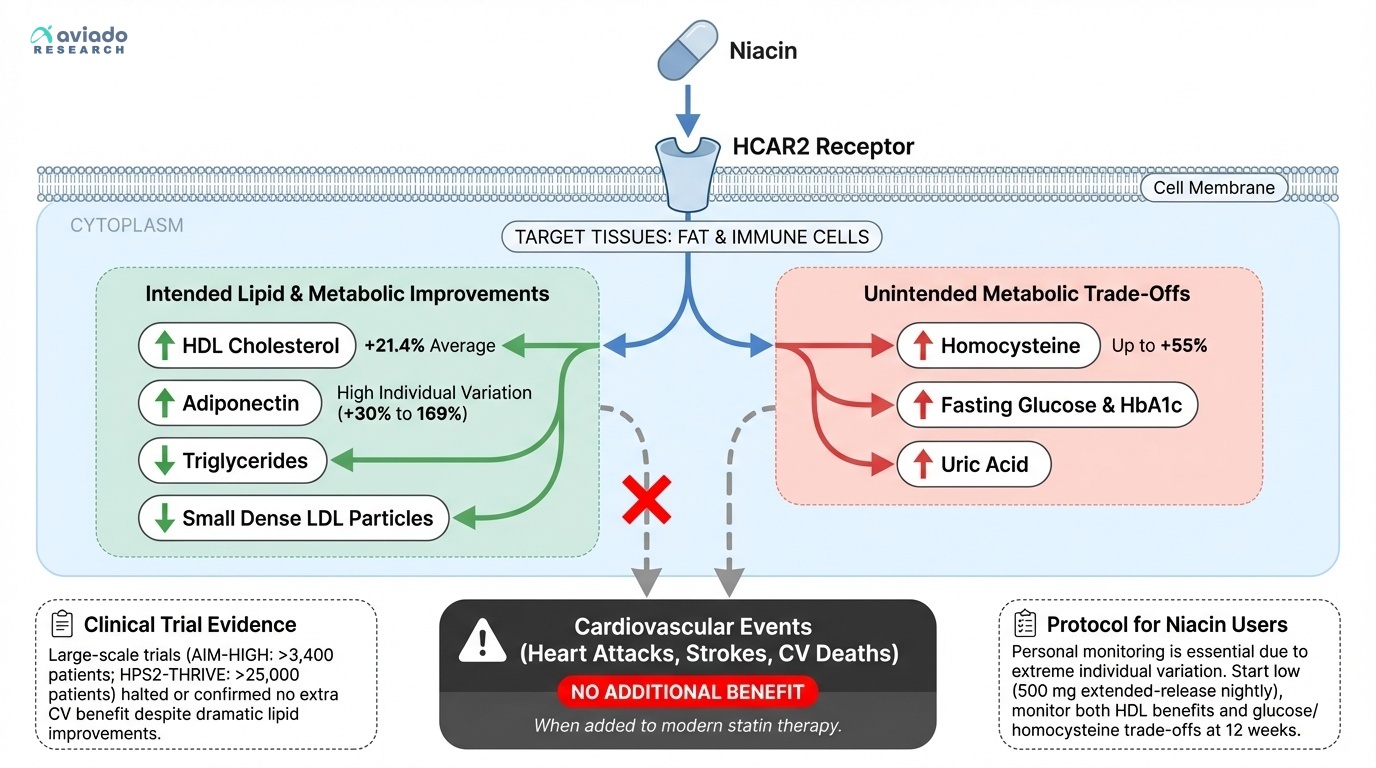
For decades, niacin was cardiologists' secret weapon. No other medication could match its ability to comprehensively improve lipid profiles—it raised HDL cholesterol, lowered triglycerides, and reduced small dense LDL particles that contribute to arterial plaque. A meta-analysis of 13 randomized controlled trials involving over 35,000 participants confirmed niacin's impressive track record: it increased HDL cholesterol by 21.4% on average [1].
But when researchers conducted large-scale trials to see if these spectacular lab improvements actually prevented heart attacks, the results were shocking. The AIM-HIGH trial, which followed over 3,400 patients, was stopped early when it became clear that adding niacin to statin therapy provided no additional cardiovascular benefit. The even larger HPS2-THRIVE trial, with over 25,000 participants, confirmed the disappointing news: despite dramatically improving cholesterol numbers, niacin didn't reduce heart attacks, strokes, or cardiovascular deaths when added to modern statin treatment [2].
This paradox reveals a fundamental problem in how we think about health optimization. Niacin works exactly as advertised on paper—it activates the HCAR2 receptor on fat cells and immune cells, triggering a cascade of beneficial metabolic changes. Yet these biochemical improvements don't necessarily translate to the clinical outcomes we actually care about.