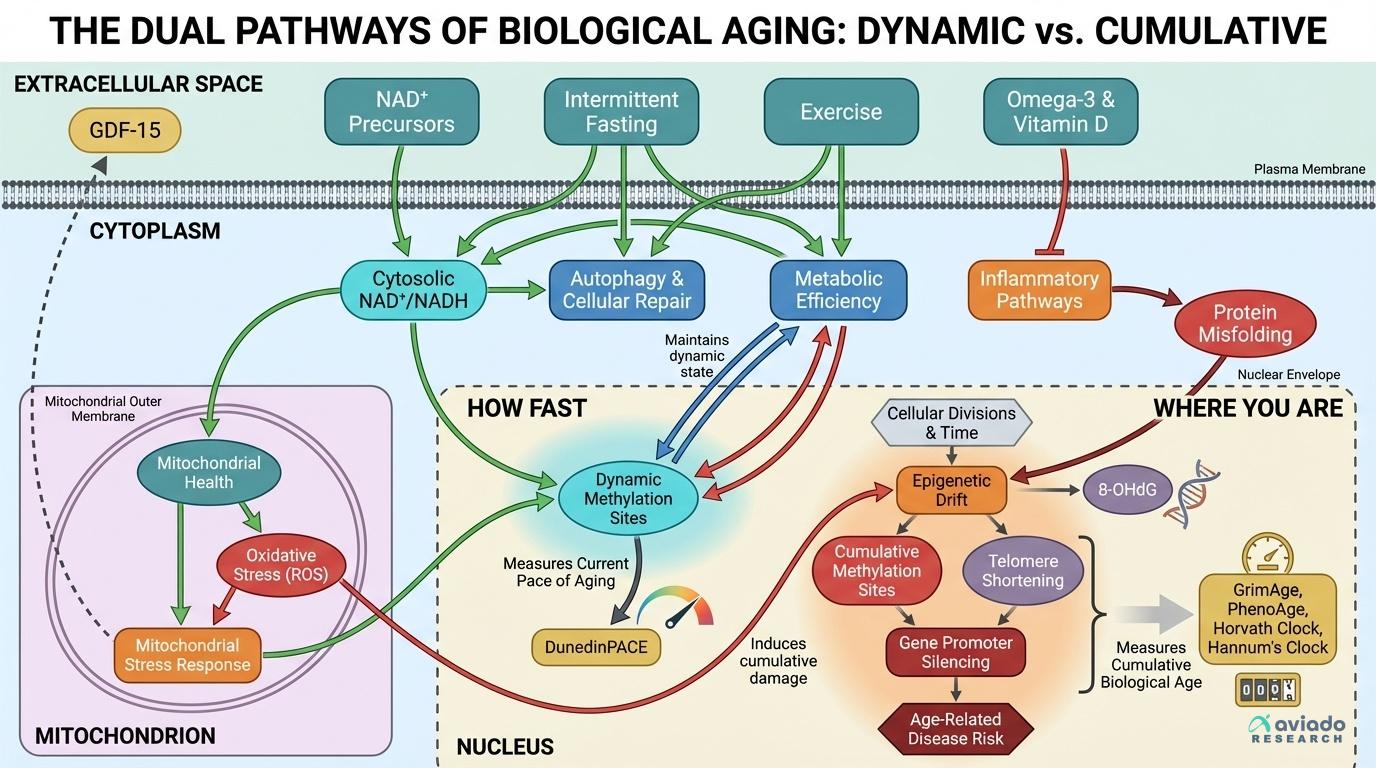
Every epigenetic aging test you can buy today is either measuring WHERE you are on the aging spectrum or HOW FAST you're currently aging — and confusing these two produces completely wrong conclusions about whether your interventions are working. Understanding this single branch point unlocks why the same supplement can 'slow aging' on one clock and show no effect on another.
The confusion stems from how DNA methylation patterns change with age. Some methylation sites accumulate damage steadily over decades, creating a biological odometer that tracks cumulative wear. Other sites respond dynamically to current metabolic conditions, functioning more like a real-time speedometer of aging velocity. First-generation clocks like Horvath's measure your total biological mileage — how much aging has already occurred. Newer clocks like DunedinPACE track your current pace of aging — whether you're accelerating or decelerating the process right now.
This distinction determines everything about how to interpret results. If your intervention slows aging velocity but you've already accumulated significant biological damage, pace-of-aging clocks will show improvement while cumulative-damage clocks may remain unchanged. The key insight: you need both measurements to understand whether you're successfully intervening in the aging process.