Magnesium L-Threonate for Brain Fog and Focus: What the Evidence Shows
Magnesium L-threonate is the magnesium form with the strongest brain-targeted rationale, and human evidence has firmed up materially.
Two independent rodent labs show it raises brain magnesium where ordinary salts do not (preliminary mechanism). Two human RCTs report cognitive gains. A 6-week self-check at the studied dose is the most useful next step.
Magnesium L-Threonate Is the Only Form Shown to Cross the Blood-Brain Barrier — But Does It Actually Work for You?
A 2024 study showed 7.5 years of cognitive age reduction in 6 weeks, but conflicting sleep results reveal why responder status matters more than average effects
Diagram glossary
- BBB:
- A highly selective membrane that regulates the passage of substances into the brain.
- citrate:
- A salt or ester of citric acid, commonly used to improve mineral supplement absorption.
- glycinate:
- A salt or ester of the amino acid glycine, often used in mineral supplements.
- PMID:
- A unique reference number assigned to every article published in the PubMed database.
- PSQI:
- A self-report questionnaire used by researchers to assess sleep quality over a one-month interval.
- threonate:
- A metabolite of vitamin C used in supplements to enhance brain magnesium delivery.
- Magnesium L-threonate
- A magnesium salt where the carrier ion is L-threonate, a metabolite of vitamin C; engineered to raise brain magnesium more effectively than standard magnesium forms.
- Blood-brain barrier (BBB)
- The selective filter formed by brain endothelial cells that controls which substances can enter brain tissue from the blood; excludes most ionic compounds including standard magnesium salts.
- Elemental magnesium
- The pure magnesium content of a supplement, separate from the carrier compound. 2,000 mg of magnesium L-threonate provides ~144 mg of elemental magnesium.
- Synapse
- The junction where one neuron transmits a signal to another; synapse density is one mechanism by which brain magnesium may support learning and memory.
- NMDA receptor
- A glutamate receptor central to learning, memory, and pain signaling; magnesium acts as a physiological modulator at this receptor and is part of why brain magnesium status matters.
- Magnetic resonance spectroscopy (MRS)
- An MRI-based imaging technique that can quantify brain magnesium concentration noninvasively; currently research-grade rather than routine clinical care.
- Magnesium glycinate
- Magnesium bound to the amino acid glycine; gentle on the stomach, widely used for sleep latency and general repletion; does not preferentially raise brain magnesium.
- Magnesium citrate
- Magnesium bound to citric acid; cheap per mg elemental; preferred for general repletion when bowel tolerance allows; does not preferentially raise brain magnesium.
- Pittsburgh Sleep Quality Index (PSQI)
- A validated self-report instrument used in many sleep RCTs; its insensitivity to short-duration interventions is a noted limitation.
- Responder vs non-responder
- The clinical recognition that any given intervention helps some individuals and not others; the magnesium L-threonate evidence base shows substantial responder spread.
- Cognitive age
- A descriptive translation of test scores into age-equivalent norms; a communication shortcut, not a measure of biological aging.
- 6-week self-check
- A structured personal-experiment framework: baseline → intervention at studied dose → planned reassessment with predefined criteria for continuing or discontinuing.
Does magnesium L-threonate cross the blood-brain barrier?
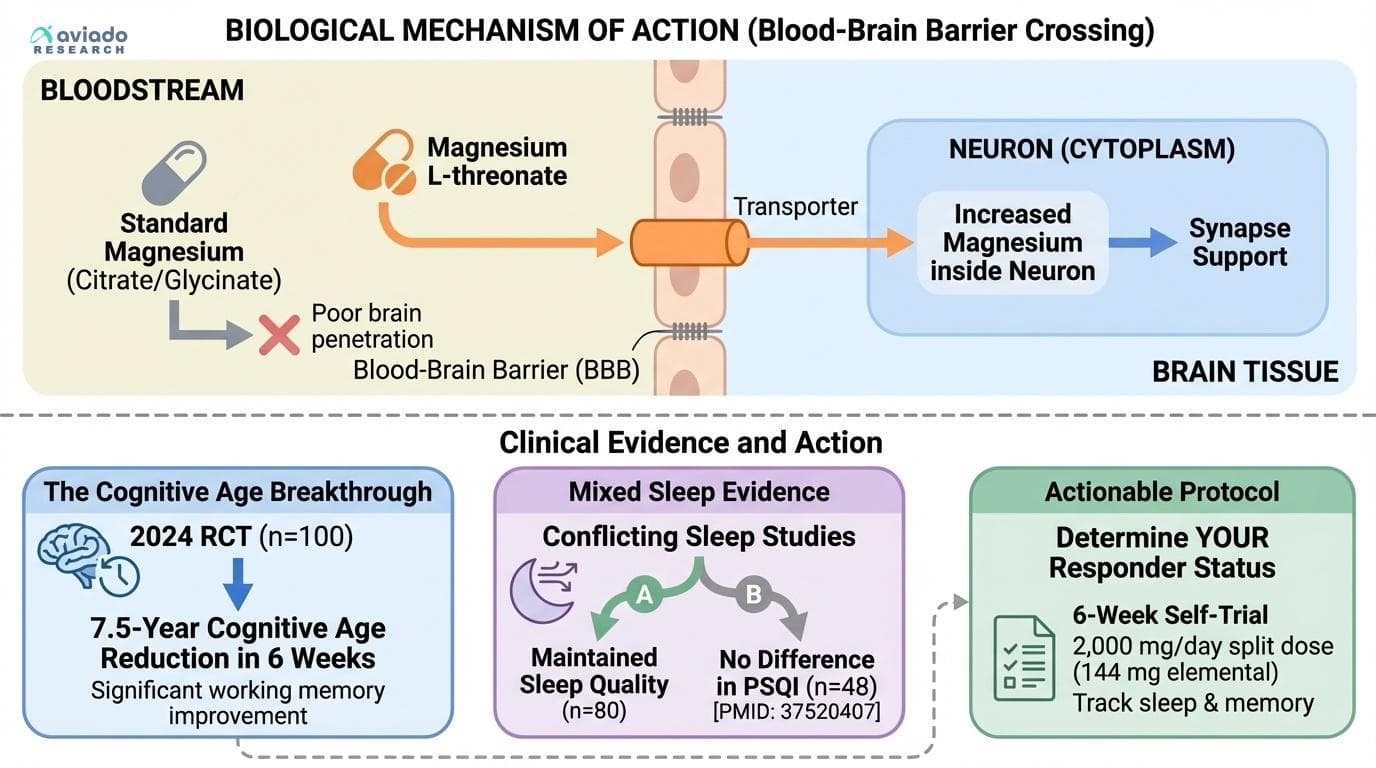
Magnesium L-threonate is the only magnesium form with replicated rodent evidence of crossing the blood-brain barrier and raising brain magnesium — and the finding has now been independently confirmed in two separate labs, with different disease models.
Think of the brain like a fortified inner city. The blood-brain barrier (BBB) functions like customs: it inspects what crosses, lets some compounds pass freely, and excludes most ionic forms — including ordinary magnesium salts. Citrate and glycinate are well-supported for raising blood magnesium and general magnesium status, but the BBB largely keeps them out of brain tissue. That is why a perfectly normal serum magnesium reading can coexist with brain-tissue magnesium that is functionally low.
Magnesium L-threonate was specifically engineered around this constraint. The threonate carrier — a sugar-acid metabolite of vitamin C — appears to act as a "shipping label" the BBB recognizes, escorting the magnesium ion across in a way other forms do not. In the foundational rodent work, oral magnesium L-threonate raised cerebrospinal fluid and hippocampal magnesium levels measurably; equivalent oral doses of magnesium chloride or magnesium gluconate did not (Slutsky et al., Neuron, 2010). Synapse density measures and long-term potentiation improved in a dose-dependent way. Subsequent mechanistic work (Sun et al., Neuropharmacology, 2016) confirmed the synapse-density signal and traced it to intraneuronal magnesium concentration.
A second, independent rodent confirmation came from a different lab and a different disease model. Shen et al. (Neuropsychiatric Disease and Treatment, 2019) directly measured cerebrospinal fluid magnesium after 4 weeks of oral dosing in mice: MgT raised CSF magnesium; oral magnesium sulfate did not — replicating the form-specificity of the original Slutsky finding. This is the key replication that lifts the BBB-crossing claim from "one paper" to "two independent rodent demonstrations of CSF-magnesium elevation specific to the threonate carrier."
Fu et al. (Brain Research, 2024) extended the BBB-relevant work in a mouse model of neuromyelitis optica spectrum disorder, showing long-term magnesium L-threonate supplementation supported BBB integrity, reduced neuroinflammation markers, and slowed demyelination compared to controls. The disease model is specific, but the BBB-integrity finding is mechanistically informative beyond the model context — it suggests threonate's relationship with the barrier may be bidirectional.
A complementary mechanism may also contribute. Liao et al. (Neural Regeneration Research, 2023) reported that magnesium L-threonate in an APP/PS1 Alzheimer's mouse model modulated the gut microbiota and repaired intestinal tight-junction proteins (ZO-1, occludin, claudin-5). This suggests part of the cognitive signal in mouse studies may route through the gut-brain axis — a parallel pathway to direct BBB transit, not a substitute for it.
Human evidence for direct brain-magnesium elevation rests on indirect inference. Cognitive RCTs (next section) show effects consistent with brain magnesium increase. No widely-available human magnetic resonance spectroscopy (MRS) study has yet directly measured brain magnesium before and after threonate supplementation in a placebo-controlled design. A definitive human imaging confirmation remains an open trial design opportunity.
The BBB-crossing claim is animal-replicated, human-suggestive. Treat it as a mechanistic anchor for why this form might matter, not as a settled human fact.
How L-threonate compares to glycinate and citrate for brain-targeted goals:
| Form | Best supported use | Brain delivery | Notes | |---|---|---|---| | L-Threonate | Brain magnesium support — cognition, sleep protection, NMDA-related uses | High (animal-confirmed; human-suggestive) | The BBB-crossing form; expensive per mg elemental | | Glycinate | Sleep latency, general magnesium repletion | Low | Gentle on stomach; well-tolerated; widely studied for sleep | | Citrate | Bowel regularity, general magnesium repletion | Low | Cheapest per mg elemental; osmotic effect is the limiting factor at higher doses |
The practical distinction: if the goal is brain effects, L-threonate is the form with the strongest mechanistic case and the only form with direct cognitive RCT evidence. If the goal is sleep latency without specific brain-magnesium concern, glycinate has a longer track record and is substantially cheaper per night. The forms are not interchangeable for brain endpoints. Substituting glycinate for threonate in a brain-magnesium protocol is not "the same intervention with cheaper math" — the BBB-crossing property is what makes threonate biologically distinct, and elemental-magnesium parity at the swallow does not translate to magnesium parity at the synapse.
Stacking is common — many users pair threonate (brain) with glycinate (sleep) at lower doses of each, particularly when the threonate is taken AM + early-PM and a smaller glycinate dose is taken at bedtime.
---
What do the human studies show?
What do magnesium L-threonate studies show? Two independent human RCTs report cognitive gains — across different populations, different cognitive batteries, and similar protocols — meaningfully increasing confidence that the cognitive signal is real.
The 2026 healthy-adult trial (Lopresti AL & Smith SJ, Frontiers in Nutrition, 2026; DOI: [10.3389/fnut.2025.1729164](https://doi.org/10.3389/fnut.2025.1729164)) enrolled 100 healthy adults aged 18–45 with self-reported sleep dissatisfaction. The intervention arm took 2 g/day Magtein magnesium L-threonate over 6 weeks; controls took placebo. The trial used the NIH Cognitive Toolbox plus Raven's Progressive Matrices for cognition, self-report sleep questionnaires, a reaction-time test, and an Oura Ring for objective sleep tracking.
Reported results:
- Cognition: Magtein outperformed placebo on the NIH Total Cognition Composite (p=0.043), with the largest treatment effects on working memory and episodic memory. - Reaction time: improved on Magtein (p=0.031). - Cognitive age: estimated brain cognitive age reduced by 7.5 years (a descriptive translation of test-score deltas to age-equivalent norms — not biological aging reversal). - Raven's Progressive Matrices: no group difference (p=0.953) — the cognitive improvements were not uniform across all measures. - Self-report sleep: Magtein improved sleep-related impairment (p=0.043) but not sleep disturbances or restorative sleep in the full sample. A subset with more severe baseline sleep problems showed a sleep-disturbance benefit (p=0.031) — direct evidence of responder spread. - Objective sleep (Oura): no group differences in measured sleep outcomes. - Autonomic markers: resting heart rate decreased (p=0.030) and heart rate variability increased (p=0.036).
The 2016 MCI trial (Liu et al., Journal of Alzheimer's Disease, 2016; DOI: [10.3233/JAD-150538](https://doi.org/10.3233/JAD-150538)) was a 12-week randomized double-blind placebo-controlled trial in older adults aged 50–70 with cognitive impairment (n=44; MgT n=23, placebo n=21). Overall cognitive ability improved significantly on MMFS-01 versus placebo (p=0.003; Cohen's d ≈ 0.91), with the largest effect on executive function — the trial population had measurably more severe executive deficits than age-matched controls at baseline, and MgT nearly restored that domain.
The two trials together — different populations (healthy young/middle-aged vs. cognitively impaired older), different cognitive batteries, similar protocol direction (~2 g/day MgT, 6–12 weeks), same direction of cognitive effect — meaningfully increase confidence that the cognitive signal is real and not single-trial noise.
The opioid-sparing trial adds independent functional confirmation. A 2023 randomized double-blind placebo-controlled trial in advanced cancer patients (Wu et al., Cancer Medicine, 2023; n=83; DOI: [10.1002/cam4.4922](https://doi.org/10.1002/cam4.4922)) showed that magnesium L-threonate supplementation (1.5–2 g daily) was associated with smaller daily morphine dose escalations versus placebo — roughly 50% lower opioid escalation rate sustained out to three months. Magnesium modulates NMDA receptor signaling, a pathway central to pain processing; low brain magnesium can amplify central pain sensitization. The cancer-pain trial adds independent evidence that brain-magnesium changes function have clinical signal beyond memory and sleep.
The sleep evidence reveals responder spread. In a 21-day randomized double-blind trial in adults aged 35–55 with self-assessed sleep problems (Hausenblas et al., Sleep Medicine: X, 2024; n=80; 1 g/day MgT), MgT outperformed placebo on both objective Oura measures and subjective measures (mood, daytime energy, mental alertness). A third sleep-relevant trial — Ni et al. (Breast Cancer (Dove Med Press), 2023; DOI: [10.2147/BCTT.S413435](https://doi.org/10.2147/BCTT.S413435)) in 109 post-mastectomy patients — showed no significant differences on PSQI, GAD-7, PHQ-9, or cognition. The trial population (post-cancer-surgery patients with persistent neuropathic pain) is so different from healthy adults that interpreting it as a "sleep RCT in general" is overreach; it establishes that MgT is not a universal intervention across all clinical contexts.
The divergence across trials is most simply explained by population differences and real responder spread. Trial averages mask the distribution — the Lopresti subset finding directly demonstrates it.
What the human evidence still does not establish: whether 2026 findings generalize to older adults with normal cognition; whether effects persist beyond 6 weeks; APOE4-stratified response; MRS imaging confirmation of brain-magnesium elevation in the same trial design; direct head-to-head against glycinate at matched elemental-magnesium doses.
Honest evidence ladder:
| Claim | Evidence tier | Anchor | |---|---|---| | Threonate raises brain/CSF magnesium where other forms don't | Animal-replicated (two independent rodent demonstrations) | Slutsky et al., Neuron, 2010; Shen et al., Neuropsychiatr Dis Treat, 2019 | | BBB integrity support with chronic supplementation | Single mouse model (NMOSD context) | Fu et al., Brain Research, 2024 | | Gut-brain axis contribution (parallel mechanism) | Single animal study | Liao et al., Neural Regen Res, 2023 (APP/PS1 mouse model) | | Cognitive performance benefit in healthy adults | Single small RCT | Lopresti & Smith, Front Nutr, 2026 (n=100, ages 18–45, 6 weeks); awaits independent replication | | Cognitive performance benefit in MCI | Single small RCT | Liu et al., J Alz Dis, 2016 (n=44, ages 50–70, 12 weeks) | | Sleep — subjective improvement / protection | Mixed small RCTs across populations | Hausenblas 2024 (signal); Lopresti 2026 (subset signal); Ni 2023 (no signal in cancer-surgery pop) | | Opioid-sparing in advanced cancer pain | Single trial in specific population | Wu et al., Cancer Med, 2023 (n=83; sustained ~50% lower opioid escalation through day 90) | | Brain fog in healthy adults at baseline | Plausibility only | No direct RCT; mechanism + responder reasoning |
All findings above are preliminary. Neither human RCT has been independently replicated, and neither was designed to confirm direct brain-magnesium elevation via imaging.
---
Can magnesium L-threonate help brain fog, focus, and memory?
Can magnesium L-threonate help brain fog? The cognitive signal across the two human RCTs is real but requires careful reading — neither trial enrolled participants specifically for "brain fog" as a primary endpoint, and the effect sizes vary by domain.
Brain fog has no direct RCT evidence as a primary endpoint for magnesium L-threonate. The mechanistic case is plausible — slowed processing and mental fatigue under load can be magnesium-responsive, and the 2026 cognitive RCT (Lopresti & Smith) showed working-memory and reaction-time gains that overlap substantially with the brain-fog phenotype. If your cognitive complaints are primarily slowed processing, difficulty sustaining attention under load, and word-finding difficulty — especially when baseline diet is magnesium-low — the responder probability is higher than average based on the available evidence.
For focus and working memory specifically, the 2026 trial showed improved overall cognition on the NIH Total Cognition Composite (p=0.043), with the largest effects on working and episodic memory and reaction time, in healthy adults aged 18–45 at 2 g/day over 6 weeks (Lopresti & Smith, Front Nutr, 2026). Notably, the same trial showed no group difference on Raven's Progressive Matrices — improvement was domain-specific, not a uniform lift across all cognitive tasks.
For memory specifically in cognitively-impaired older adults, the MCI trial (Liu et al., J Alz Dis, 2016) showed broad cognitive improvement (MMFS-01, p=0.003; Cohen's d ≈ 0.91) after 12 weeks, with the largest effect on executive function. Memory endpoints improved significantly in the MCI population.
Sleep spread as a responder signal. The sleep heterogeneity story is also informative for the brain-fog question. The 2026 Lopresti trial found that the subset of participants with more severe baseline sleep problems was the subgroup that most clearly benefited — which is consistent with the broader pattern across all the trials: response concentrates in those who have a relevant baseline deficit, whether that is magnesium status, sleep quality, or cognitive load.
The brain neurometabolism angle adds further context. Estrogen withdrawal during the perimenopausal-to-early-postmenopausal window is a neurometabolic event in its own right — cerebral glucose metabolism declines and mitochondrial cytochrome-oxidase activity drops in AD-vulnerable regions during this transition, with a gradient most pronounced in postmenopause (Mosconi et al., PLOS One, 2017; DOI: [10.1371/journal.pone.0185926](https://doi.org/10.1371/journal.pone.0185926)). The cognitive symptoms many women report in this transition overlap substantially with the cognitive phenotype that magnesium L-threonate is studied for. Direct trial evidence in perimenopausal women specifically does not yet exist, but the responder profile may concentrate in this population given the simultaneous shift in magnesium balance and brain neurometabolism.
The key calibration: single-trial evidence in each population is not the same as established benefit, and individual response varies substantially. Treat brain-fog and focus improvement as plausible for the studied dose with a 6-week trial duration; do not expect dramatic effects within the first two weeks. If you have untreated sleep apnea, thyroid dysfunction, or significant medication side effects, those are higher-yield targets than supplementation.
A note on dosing. Many magnesium L-threonate products list 2,000 mg "magnesium L-threonate" per serving but only ~144 mg "elemental magnesium." Both numbers are correct — the first is the full magnesium-threonate compound, the second is the magnesium portion separated from the threonate carrier. The therapeutic effect appears tied to the threonate-mediated brain delivery, not to the elemental magnesium count, which is why direct dose comparisons against other magnesium forms are not straightforward.
---
Who is most likely to respond, and how to run a 6-week self-check?
Who is most likely to respond to magnesium L-threonate? Responder spread across the trials suggests response concentrates in adults who have a relevant baseline deficit — whether in magnesium status, cognitive load, or sleep quality. Plausible responder profiles based on the available evidence:
- Adults whose cognitive complaints include slowed processing, mental fatigue under load, or word-finding difficulty, particularly when baseline diet is magnesium-low - Adults with sleep complaints where magnesium status may be a contributor (rule out sleep apnea, anxiety, circadian misalignment first) - Older adults experiencing age-typical working-memory slowing - People with high cognitive demand under stress or sleep restriction (the brain-magnesium signaling pathways are most strain-sensitive) - Possibly: women navigating the perimenopausal-to-postmenopausal transition, where magnesium balance shifts and brain neurometabolism changes simultaneously — though direct trial evidence in this population is currently limited (Mosconi et al., PLOS One, 2017)
If you want to test whether you personally respond, a structured 6-week self-check is the most evidence-grounded approach (informed by the Lopresti & Smith 2026 trial duration):
1. Establish a baseline. Track 2 weeks of nightly sleep quality (a 1–10 self-rating or a wearable's deep-sleep minutes) and weekly cognitive performance (a brief validated test like Cogstate, BrainCheck, or simple word-recall) before starting. Write down what you would consider a meaningful change — defining the success criterion before you start prevents motivated interpretation later. 2. Run the protocol. 2,000 mg magnesium L-threonate daily, split AM + PM, for 6 weeks. Continue baseline tracking nightly (sleep) + weekly (cognition). Hold all other variables constant where you can — adding a new exercise routine or starting a meditation practice in the same window will confound your read. 3. Compare weeks 4–6 against 0–2. Look for changes meaningful to you, not trial-grade statistical significance — your trial is N=1. Responders typically notice changes by weeks 3–4; sustained or growing effects through weeks 5–6 increase confidence that the effect is real and not a regression-to-the-mean artifact. 4. Decide. If clear positive changes emerge, continue at the same dose; periodically test holds (a 2-week off-period after several months) to confirm the change is the supplement and not background drift. If nothing changes after 6 weeks at full dose, you are likely a non-responder for cognition — extending duration further is unlikely to help.
If you prefer not to run a structured self-check, the same cost-benefit logic still applies: the evidence supports a reasonable trial for those in plausible responder profiles, without a formal tracking protocol. The self-check is an option for those who want the clearest read on personal response, not a prerequisite for considering the supplement.
Cost honesty. At 2,000 mg/day, branded magnesium L-threonate runs $30–$60/month at consumer pricing. Generics are emerging but often poorly characterized — the threonate carrier complex matters, and "looks like the same molecule" is not always the same molecule biologically. If a 6-week self-check (or an informal trial period) produces no detectable change, the cost saved by discontinuing is meaningful and recurring.
What to do alongside. The highest-yield non-supplement levers for cognition remain consistent sleep (7–8 hours, OSA screened if any indicators), weekly aerobic exercise (≥150 min/week moderate intensity), blood-pressure control, and Mediterranean dietary pattern. The supplement trial is a small layered question on top of the foundation. If your foundation is shaky, the trial is unlikely to produce dramatic results regardless of responder status.
Sex-stratified considerations. The published magnesium L-threonate trials have not been powered to detect sex-by-treatment interactions. Women navigating the perimenopausal transition who are considering a trial should anchor it in the broader hormonal-cognitive picture. Address sleep architecture, vasomotor disruption of sleep, and cardiovascular risk modification first; treat the magnesium trial as an addition rather than a substitute. The same caveat applies throughout: averages are not destinies, and the absence of sex-stratified data is itself a limitation worth keeping visible until larger trials report.
---
How magnesium L-threonate fits into a broader cognitive support stack
How does magnesium L-threonate fit in a broader cognitive stack? It is one Tier 3 lever in a broader cognitive longevity protocol — defensible, mechanistically anchored, but not a structural foundation. The framework below grades each candidate intervention by evidence quality.
Tier 1 (the structural foundation — strong RCT evidence): aerobic exercise, hearing-aid use for treated hearing loss, blood-pressure control, glycemic control, sleep adequacy and OSA treatment, social engagement. These are the load-bearing interventions; nothing in Tier 2 or Tier 3 substitutes for them.
Tier 2 (observational with biological plausibility, mixed RCT): Mediterranean dietary pattern, omega-3 fatty acids (especially DHA), B-vitamins for elevated homocysteine. These layer onto the Tier 1 foundation.
Tier 3 (small trials + mechanistic plausibility): magnesium L-threonate, Lion's Mane mushroom extract, magnesium in other forms for sleep, bioavailable curcumin, creatine monohydrate. Tier 3 interventions are reasonable additions for those who want depth, but they are not the high-leverage moves and should not displace Tier 1 work.
Tier 4 (genotype-personalized): APOE4-stratified protocols including higher omega-3 dosing in carriers, saturated-fat caution, and exercise-intensity tuning.
The cluster relationship. Magnesium L-threonate addresses a specific, narrow biological lever — brain-magnesium availability — that may or may not be a binding constraint in your case. The 6-week self-check (structured or informal) determines whether it is. If you are a responder, it integrates well alongside the broader protocol; if you are not, the protocol's Tier 1 work is what is doing the actual prevention. Both outcomes are useful information.
For the broader magnesium picture beyond brain-targeted use, see [why your magnesium supplement may be working — just not where you're looking](/research/articles/why-your-magnesium-supplement-may-be-workingjust-not-where-youre-looking/), which covers magnesium's better-established cardiovascular and metabolic roles. For the magnesium ingredient hub across forms, see the [Magnesium research hub](/research/supplements/magnesium/). For brain-targeted supplement context, the [Magnesium L-threonate (brain-targeted form)](/research/supplements/magnesium-threonate/) page covers dosing, sourcing, and form-selection in more detail. The cognitive longevity framework is developed in full in the [Brain, Mood & Cognitive Performance pillar](/research/pillars/brain-cognitive/).
---
Conclusions
Magnesium L-threonate targets brain magnesium rather than general magnesium status, and that biological distinction is the article's reason for being. The BBB-crossing claim is now anchored in two independent rodent demonstrations (Slutsky 2010; Shen 2019), not one. Two human cognitive RCTs in different populations report cognitive improvement (Liu 2016 in MCI; Lopresti & Smith 2026 in healthy adults) — the strongest evidence base this form has had to date, though both trials remain small and neither has been independently replicated. The headline "7.5-year cognitive age reduction" from the 2026 trial warrants careful reading: it is a descriptive translation of test-score deltas, not biological aging reversal. Sleep findings are mixed across populations and doses, with the Lopresti subset finding directly demonstrating responder spread. The opioid-sparing trial in advanced cancer patients (Wu et al., 2023) provides independent evidence that brain-magnesium changes function in clinically meaningful ways via a non-self-reported endpoint. The most practical use of the current evidence is a trial at the studied dose with explicit responder vs. non-responder framing — structured or informal — rather than indefinite supplementation based on average trial effects. Evidence tier: preliminary. This article will be updated as larger replications and head-to-head form-comparisons report.
Several constraints apply. (1) Human evidence remains modest — the cognitive RCT base consists of two small trials in different populations (Liu 2016 in MCI, n=44, 12 weeks; Lopresti & Smith 2026 in healthy adults, n=100, 6 weeks). Same-team independent replication of either result has not been published. (2) Brain-penetration claims rely on rodent work; no widely-available human MRS study has yet directly measured brain magnesium before and after threonate supplementation in a placebo-controlled design. (3) Sleep evidence conflicts across trials and populations, with subjective measures often improving while objective Oura measures are mixed; head-to-head designs that pre-stratify likely responders are absent. (4) The "7.5-year cognitive age" framing translates test-score deltas to age-equivalent norms; it is a useful description, not a measure of biological aging reversal. (5) Direct head-to-head trials comparing magnesium L-threonate against glycinate or citrate at matched elemental-magnesium doses are essentially absent — practical form selection rests on mechanistic inference. (6) Long-term safety beyond ~12 weeks at 2,000 mg/day is not directly studied; magnesium has a wide therapeutic margin in healthy adults but renal-impaired individuals should not supplement without clinician guidance. (7) Sex-stratified data is essentially absent across the trial base; the perimenopausal-cognitive responder hypothesis is plausibility, not data. Specific clinical decisions belong with the patient and their clinician.
Frequently Asked Questions
What are the benefits of magnesium L-threonate?
The strongest human signals are cognitive performance improvement in healthy adults over 6 weeks at 2 g/day (Lopresti & Smith, *Front Nutr*, 2026) and cognitive improvement in older adults with mild cognitive impairment over 12 weeks (Liu et al., *J Alz Dis*, 2016). These gains rest on two independent, small RCTs — both preliminary, neither independently replicated. Sleep effects are mixed across populations and doses, with subjective improvements more consistent than objective ones. Sustained reduction in opioid escalation has been reported in advanced cancer pain (Wu et al., *Cancer Med*, 2023). The core mechanistic distinction is brain-targeted magnesium delivery via BBB crossing — a property replicated in two rodent labs — which distinguishes it from other magnesium forms for brain-specific goals.
Does magnesium L-threonate cross the blood-brain barrier?
Yes, in animal studies — and the finding has been independently replicated. Slutsky et al. (*Neuron*, 2010) showed oral magnesium L-threonate raised brain magnesium in rats where equivalent doses of magnesium chloride or gluconate did not. Shen et al. (*Neuropsychiatr Dis Treat*, 2019) replicated the form-specificity in a different lab and a different model: in mice, oral MgT raised cerebrospinal fluid magnesium directly; oral magnesium sulfate did not. Long-term mouse studies have separately shown supportive effects on BBB integrity in disease models (Fu et al., *Brain Research*, 2024). Direct human MRS imaging confirmation has not yet been published in a placebo-controlled design; human evidence is inferred from cognitive and pain trials. Treat the BBB-crossing claim as animal-replicated, human-suggestive.
Can magnesium L-threonate help brain fog?
Brain fog has no direct RCT evidence as a primary endpoint for magnesium L-threonate. The mechanistic case is plausible — slowed processing and mental fatigue under load can be magnesium-responsive, and the 2026 cognitive RCT (Lopresti & Smith) showed working-memory and reaction-time gains that overlap with the brain-fog phenotype. Responder heterogeneity is substantial: the subset of trial participants with more severe baseline deficits showed stronger gains. A trial at 2 g/day with a structured baseline — or simply a defined evaluation period — is the most evidence-grounded way to determine personal response. Rule out sleep apnea, thyroid dysfunction, and medication side effects first; those are higher-yield targets when present.
Does magnesium L-threonate improve focus or memory?
The 2026 trial showed improved overall cognition (NIH Total Cognition Composite, p=0.043), with the largest effects on working and episodic memory and reaction time, in healthy adults aged 18–45 at 2 g/day over 6 weeks (Lopresti & Smith, *Front Nutr*, 2026). Notably, the same trial showed no group difference on Raven's Progressive Matrices — improvement was domain-specific. A separate small RCT in mild cognitive impairment showed broader cognitive improvement including memory at 12 weeks (Liu et al., *J Alz Dis*, 2016). Single-trial evidence in each population is not the same as established benefit, and individual response varies. Treat as plausible for focus and working memory at the studied dose with a 6-week evaluation window; do not expect dramatic effects within the first two weeks.
What do magnesium L-threonate studies show?
The evidence ladder has three tiers. First, two independent rodent labs show it raises brain and CSF magnesium where other forms do not (Slutsky et al., *Neuron*, 2010; Shen et al., *Neuropsychiatr Dis Treat*, 2019) — the animal-replicated foundation. Second, two human RCTs report cognitive gains: one in healthy adults aged 18–45 (Lopresti & Smith, *Front Nutr*, 2026; n=100, 6 weeks) and one in older adults with mild cognitive impairment (Liu et al., *J Alz Dis*, 2016; n=44, 12 weeks). Third, three sleep trials show mixed results across different populations and doses — Hausenblas et al. 2024 (signal at 1 g/day), Lopresti & Smith 2026 (subset signal at 2 g/day), Ni et al. 2023 (no signal in post-mastectomy patients) — directly demonstrating responder spread. All human findings are preliminary and neither cognitive RCT has been independently replicated.
Is magnesium L-threonate better than magnesium glycinate or citrate?
For brain-targeted goals, magnesium L-threonate has a stronger mechanistic and direct-trial case: two independent rodent labs confirm it crosses the blood-brain barrier and raises brain magnesium where glycinate and citrate do not; two human cognitive RCTs report gains that glycinate and citrate have no equivalent evidence for in brain endpoints. For sleep latency and general magnesium repletion, glycinate has a longer track record, is substantially cheaper per mg elemental, and is well-tolerated. For general magnesium status (low dietary intake, restless leg, mild constipation), citrate or glycinate suffice. The forms are not interchangeable for brain endpoints — different forms for different goals. Direct head-to-head RCTs at matched elemental-magnesium doses for brain endpoints are essentially absent; form selection rests on goal alignment and mechanistic inference.
How long does magnesium L-threonate take to work?
In the published RCTs, cognitive effects emerged within 3–4 weeks and continued through study end. The 2026 healthy-adult trial ran 6 weeks; the 2016 MCI trial ran 12 weeks. Sleep effects, when present, appeared within the first 2–3 weeks. The practical implication: a 6-week evaluation window is the evidence-grounded minimum for cognitive endpoints — effects from the 2026 trial that span 6 weeks cannot be reliably judged at 2 or 3 weeks. Absence of any change after 6 weeks at the studied dose (2,000 mg/day) suggests non-responder status for cognition; extending duration further beyond that window is unlikely to produce results. Cognitive endpoints typically need 4–12 weeks for the brain-magnesium changes to translate into measurable functional gains. ---
Track this in your stack
See how magnesium threonate relates to your health goals and monitor changes in your biomarkers over time.
